Cell Systems and Drug Safety
Affinity
Designing and synthesizing high-affinity ligands for a given drug target have always been among the more important challenges in the drug discovery process.
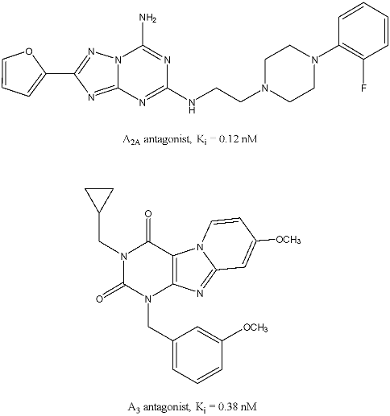
We have done that routinely over the years and have been successful on many occasions. It does make sense to have high affinity, next to other design criteria. Administering, as a consequence, low amounts of compound to a patient is often beneficial in terms of side effects, and particularly in metabolic load. Interestingly, compounds with super-high affinity (that is subnanomolar affinity) are rare, and we’re proud that we’ve managed to develop a few for adenosine and cannabinoid receptors (see Figure for a few representative examples).
Related publications
- Xia L., de Vries H., Lenselink E.B., Louvel J., Waring M.J., Cheng L., Pahlén S., Petersson M.J., Schell P., Olsson R.I., Heitman L.H., Sheppard R.J., IJzerman A.P., Structure-Affinity Relationships and Structure-Kinetic Relationships of 1,2-Diarylimidazol-4-carboxamide Derivatives as Human Cannabinoid 1 Receptor Antagonists. J Med Chem 2017, 60(23): 9545-9564.
- Xia L., Burger W.A.C., van Veldhoven J.P.D., Kuiper B.J., van Duijl T.T., Lenselink E.B., Paasman E., Heitman L.H., IJzerman A.P., Structure-Affinity Relationships and Structure-Kinetics Relationships of Pyrido[2,1-f]purine-2,4-dione Derivatives as Human Adenosine A3 Receptor Antagonists. J Med Chem 2017, 60(17): 7555-7568.
- Guo D., Xia L., van Veldhoven J.P., Hazeu M., Mocking T., Brussee J., IJzerman A.P., Heitman L.H., Binding kinetics of ZM241385 derivatives at the human adenosine A2A receptor. ChemMedChem 2014, 9(4): 752-61.

