LUMC researchers: matching medication to DNA leads to 30% fewer side effects
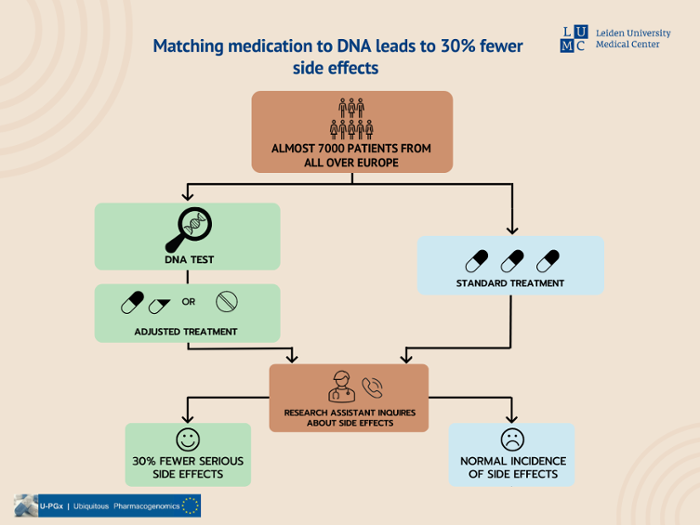
According to an international group of researchers led by Leiden University Medical Center (LUMC), patients experience 30% fewer serious side effects when medication doses are tailored to their DNA. The study, published in The Lancet, is the first to demonstrate the practical application of prescribing drugs based on an individual’s genetic information.
The ‘one-size-fits-all’ approach to prescribing medication is outdated. Due to variations in our genetic information, patients may respond differently to a specific drug. For example, some individuals process medication faster than others and, as a result, require a higher dose to achieve the necessary intervention effect. Personalised treatments are therefore desirable. To facilitate this, researchers have developed a ‘DNA medication pass’ that associates a patient’s genetic profile to drugs whose processing is influenced by DNA. Doctors and pharmacists scan the pass to find out the optimal medication dose for the individual being treated.
Standard vs. adjusted dose
The Lancet study found that patients who actively made use of the medication pass, and whose doses were adjusted to their DNA, experienced 30% fewer serious side effects than patients who were prescribed a standard dose of medication. Approximately 7000 patients from seven European countries were assessed over several medical specialities, including oncology, cardiology, psychiatry and general medicine.
All participants were prescribed a drug whose processing is influenced by our genes. First, the DNA of each patient was mapped. The researchers then looked at 12 specific genes and discovered that 50 types of genetic variant affected how the 39 selected drugs worked. Up to 12 weeks after initiating treatment, the patients were contacted by a nurse specialist who enquired about their side effects, such as diarrhoea, anaemia, nerve pain or loss of taste.
Not only did the patients with the DNA medication pass experience fewer side effects, but they also expressed great satisfaction with the pass itself. According to the researchers, the pass gives patients the feeling of being more in control, as they become actively involved in their personalised treatment.
‘We want to move towards mapping the DNA of every patient who comes to the pharmacy’
Standard care?
The study was coordinated by Henk-Jan Guchelaar, Professor of Clinical Pharmacy at LUMC. He has conducted research in the field of pharmacogenetics for over 20 years. ‘For the first time we have proven that a “tailored” strategy works at a large scale within clinical practice. There is now enough evidence for us to proceed with implementation,’ says Guchelaar. ‘This means the next step for us is to start making use of the DNA medication pass,’ adds Jesse Swen, Professor of Clinical Pharmacy and principal investigator.
Figuring out the subsequent stages of the implementation process also raises various questions for the researchers: Should the pass be reimbursed? And should it be considered part of standard care? Guchelaar and Swen believe it should. According to them, this study is good reason to do so. ‘We want to move towards mapping the DNA of every patient who comes to the pharmacy,’ says Guchelaar. ‘Then we can make treatment more effective and safer for each patient.’
This study was funded by a European Commission Horizon 2020 grant (No 668353).